Authors Guideline
Article Types
The International Journal of Pharmaceuticals, Nutraceuticals and Cosmetic Science (IJPNaCS) accepts original research articles, full length review articles, mini reviews, short communications, case reports, and letters to the editor. Manuscripts must be written in a clear and concise manner and should include structured headings (refer to the preparation of manuscript guidelines for details).
General format and length of the types of articles accepted for submission
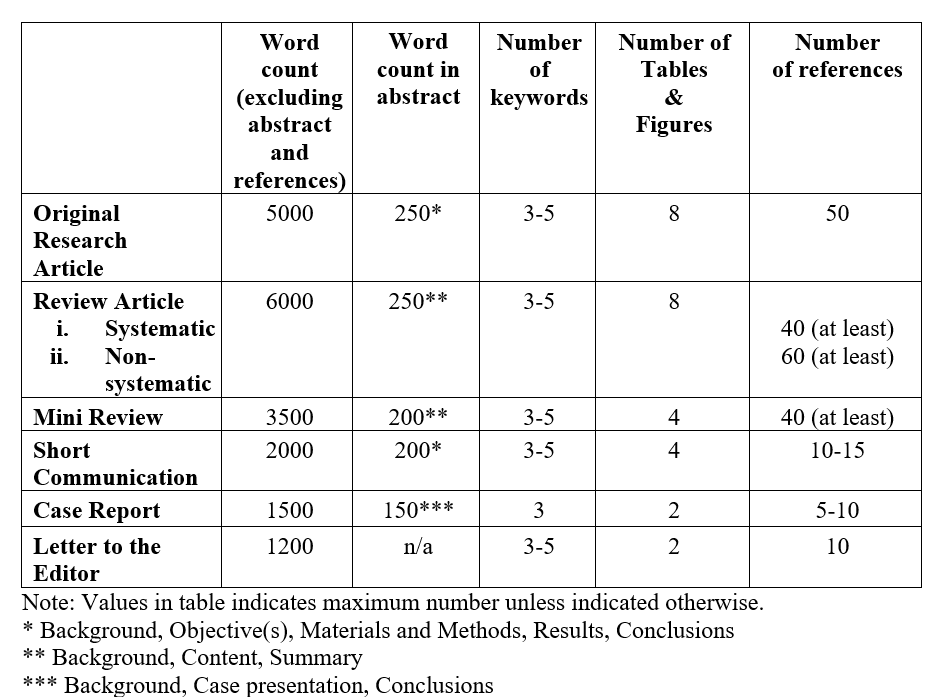
Original research articles
Articles describing substantial original research falls within the scope of the journal. Structured headings are required (refer preparation of manuscript for details).
Review articles should be prepared according to one of the following styles:
- Systematic reviews should contain original articles' parts. The protocol of these studies should adhere to PRISMA (for systematic reviews of RCTs) or MOOSE (for Observational studies) guidelines. Abstract should be structured.
- Nonsystematic reviews (Narrative reviews) should be written by experts contained the critical assessment of the current knowledge of the field. Different parts of these articles include abstract, introduction, discussion and conclusion. Abstract should be structured.
Short communication/Brief reports can be in form of research article, systematic review or ongoing research which reports its interesting findings. The parts in this type of articles are like those of original one but they are smaller and prepared in maximum 2000 words.
Case Reports: Should include abstract, keywords, case presentation, discussion, acknowledgment, references, and figures. Necessary documentations of the case(s) like pathology reports, laboratory test reports, and images should be included in the submission package.
A letter to the editor should be about criticism of previous articles, criticism or review over books, analysis of a related topic, expansion and explanation about an idea or a complicated problem. These articles need no structure.
Preparation of manuscripts
Formatting
- All manuscripts must be in English.
- Manuscripts should be single spaced; the text should be in Times New Roman 10-point font.
- Title page
- Title: The manuscript title must be center-aligned using a 17-point font. Use sentence case, capitalizing only the first letter of the first word and proper nouns. To facilitate effective information retrieval, titles should be concise and informative. Avoid the use of abbreviations, symbols, or formulae within the title.
- Author Names: Names should be centered in a 13-point font and separated from the title by a single line space.
- Corresponding Author: Clearly identify the corresponding author by placing an asterisk (*) next to their name
- Affiliations and Addresses: Full correspondence addresses should be centered, italicized, and set in an 8-point font. This section must be separated from the author names by a single line space
- Abstract
- The abstract should be a clear, concise summary (100 to 300 words with no abbreviations)—informative rather than descriptive, giving the scope and purpose, methods or procedures, significant new results, and conclusions. Use font size 9.
- 3 to 5 keywords should be provided on the title page. These words must be used in the abstract. (Use Times New Roman Font, Size 12, Italic).
- The text should normally be divided into conventional Introduction, Materials and Methods, Results, Discussion and References. Subsections should be numbered 1.1 (then 1.1.1, 1.1.2,……), 1.2, etc.
- Introduction (summarize the purpose and the rationale for the study);
- Materials and Methods (study design and exact method or observation or experiment, definitions such as for diagnostic criteria, the population or patient samples, and laboratory and statistical methods);
- Results (presented in the form of text, tables and illustrations)
- Discussion (present findings and the variations or similarities with other work done in the field by other workers)
- Conclusion (final result that the author(s) has (have) reached)
- Acknowledgements/fundings (All contributors who do not meet the criteria for authorship should be covered in the acknowledgement section. Financial and material support should also be acknowledged)
- Conflict of interest
- Authors contributions, and
- References.
Article Manuscript Submission Template For submitting article, please download the template at IJPNaCS UiTM Journal Template 2026 updated.docx
Statistical analysis
Experimental design, subject selection and randomization procedures should be described, and analytical precision quoted when appropriate. Appropriate power calculations for the sample size used should be given. In case–control studies clearly define how cases and controls were selected and what matching has taken place. Authors should detail how they have addressed missing data and loss to follow up. Analytical methods used to account for sampling strategy should be described. Rounded figures are easier to compare, and extra decimal places should be avoided. Descriptive statistics require an additional digit to those used for the raw data. Percentages should not be expressed to more than one decimal place and should not be used at all for small samples.
Normally distributed data should be described using a mean, SD and/or %CV and expressed as ‘mean (SD)’ or ‘mean ± SD’. When data are not normally distributed, then medians and interquartile ranges should be used in place of mean and SD. Skewed data can often be normalized by logarithmic transformation or a power transformation. The statistical analysis and calculation of summary statistics should be carried out on the transformed data and the summary statistics transformed back to the original scale for presentation. If a logarithmic scale is used, then graphs should display non-transformed data on a logarithmic scale. The conventional use of statistical significance is P≤0.05. If a different significance level needs to be used, then the reasons why must be clearly stated in the statistical method section.
Abbreviations and units
Abbreviations: Define abbreviations and acronyms the first time they are used in the text, even after they have been defined in the abstract. Symbols and abbreviations should be those currently in use. Authors should not create new abbreviations and acronyms. Do not use abbreviations in the title or headings unless they are unavoidable.
Units: All measurements should be expressed in SI units.
Artwork, figures and other graphics
Illustrations, pictures and graphs, should be supplied with the highest quality and in an electronic format that helps us publish your article in the best way possible. Please follow the guidelines below to enable us to prepare your artwork for the printed issue as well as the online version. Rasterized based files (i.e. with .tiff or .jpeg extension) require a resolution of at least 300 dpi (dots per inch). Line art should be supplied with a minimum resolution of 800 dpi.
Tables
All tables should be numbered with Arabic numerals. Headings should be placed above tables, left justified, and bold with font size of 8. Leave one line space between the heading and the table, by choosing Table Caption in the Styles Gallery. Only horizontal lines should be used within a table, to distinguish the column headings from the body of the table, and immediately above and below the table. Tables must be embedded into the text and not supplied separately.
Figures
Should be in limited numbers, with high quality artwork and mounted on separate pages. The captions should be placed below the figures. The same data should not be presented in tables, figures and text, simultaneously. Insert figures after they are cited in the text. Use the abbreviation “Fig.1”, even at the beginning of a sentence. Diagrams can be in the form of framework, process flow, chart, or any visual aids that are meant to support the written text. Any diagrams built using Microsoft Word or Microsoft Power Point’s Smart Art Graphic Tool needs to be embedded as editable or vector-based objects (instead of image objects). Diagrams attached using bitmap-based images (jpeg., png., and other formats) need to be in high resolution (at least 300 pixels per inch). The font used for labels or indicators inside the diagram needs to be formatted as Times New Roman with font size set at 9. It must not be bold, italicised, or underlined. Written permission must accompany any photograph in which the subject can be identified or any illustration that has been previously published.
Supplementary material
This journal may host additional materials online e.g. movie clips, questionnaires.
Conflict of interest and other declarations
All authors are required to declare any conflicts of interest when submitting papers for publication. Declarations of funding sources, ethical approval and acknowledgement should be included at the end of the manuscript (before references list).
- Declaration of competing interests: GKSS is an employee of XYZ Coorperation. GS provides consultative advice to ABC corporation.
- Funding: This research was funded by (please provide grant name and number)
- Ethical approval: The animal ethics committee of UiTM (UiTM CARE) approved this study (please provide number)
- Acknowledgements
Authors contribution
Each author contribution must be stated clearly reflecting each contribution to the body of the work and manuscript. Authors can refer to CRediT (Contribution Roles Taxonomy) for the detailed information about individual contributions to the work.
Declaration of generative AI in the writing process
The authors declare that [select one]:
- No artificial intelligence (AI) tools were used in the writing, editing, data analysis, image generation, or any other aspect of preparing this manuscript.
OR
- Artificial intelligence (AI) tools were used in the preparation of this manuscript as follows:
- Tool(s) used: [e.g., ChatGPT by OpenAI, Grammarly, etc.]
- Purpose of use: [e.g., language editing, grammar checking, summarisation of background literature, figure generation, etc.]
- Extent of use: [e.g., minor language refinement, idea generation support without direct content creation, etc.]
The authors affirm that they have critically reviewed and taken full responsibility for the final content, and that AI tools were not credited as authors.
Reference style
All manuscripts should be accompanied by relevant references. The Reference should provide the following information as stated in the presented models as follows:
In-text citations should follow the APA 7th edition author-date format, and the reference list should be arranged alphabetically by the author's last name.
Reference list should be single spaced with one line spaced with indentation between each entry.
Journal:
Reker, D., Blum, S. M., Steiger, C., Anger, K. E., Sommer, J. M., Fanikos, J., & Traverso, G. (2019). “Inactive” ingredients in oral medications. Science Translational Medicine, 11(483). https://doi.org/10.1126/scitranslmed.aau6753
Book:
Katzung, B. G., & Vanderah, T. W. (2021). Basic and clinical pharmacology (15th ed.). McGraw Hill.
Chapter in a book:
Westfall, T. C., & Westfall, D. P. (2011). Neurotransmission: The autonomic and somatic motor nervous systems. In L. L. Brunton, B. A. Chabner, & B. C. Knollmann (Eds.), Goodman and Gilman's the pharmacological basis of therapeutics (12th ed., pp. 171–218). McGraw-Hill Companies Inc.
E-book:
Rang, H. P., Ritter, J. M., Flower, R. J., & Henderson, G. (2020). Rang & Dale's pharmacology (9th ed.). Elsevier. https://doi.org/10.1016/C2016-0-03823-3
Internet document:
Hughes, B. (2026, April 14). The impact of AI on small-molecule drug discovery. Biopharma Dive. https://www.biopharmadive.com/news/ai-drug-discovery-trends-2026/
Newspaper article:
Robbins, R. (2026, January 15). How pharmacy benefit managers drive up drug costs. The New York Times. https://www.nytimes.com/2026/01/15/health/pbm-drug-costs.html
Conference paper from the internet/unpublished:
Chen, L., & Miller, S. (2026, April 12–15). AI-driven ligand screening for novel beta-blockers [Paper presentation]. International Conference on Pharmaceutical Sciences, Geneva, Switzerland. https://www.icps2026.org/abstracts/chen-miller
Conference proceeding:
Al-Farsi, M. (2026). Nanoparticle drug delivery systems for oncology. In T. R. Gupta (Ed.), Proceedings of the 14th Global Pharmacology Summit (pp. 45–58). Academic Press. https://doi.org/10.1016/B978-0-12-8192026.00012-3
Theses (full text database):
Singh, R. (2026). Pharmacokinetic modeling of sustained-release insulin in pediatric populations [Doctoral dissertation, University of Oxford]. Oxford University Research Archive. https://ora.ox.ac.uk/objects/uuid:12345-6789
Ethical conduct of research involving human and animals
The research that involves human subjects must adhere to the principles of the ethical standards of the responsible committee on human experimentation (institutional and national) and World Medical Association Declaration of Helsinki. All research participants should be informed about the aims of the study and any possible side effects of the drugs and intervention.
When reporting experiments on animals, authors should indicate whether the institutional and national guide for the care and use of laboratory animals was followed. Written informed consent under protocols approved by an institutional or local review board or approved animal protocols are essential if the research involves human or animal subjects, respectively. This information should be stated in the manuscript and the protocol number or exempt status of approved protocols should be stated in the manuscript at the time of submission for review.
Ethical considerations must be clearly addressed in the Declaration or the Methods section, and the name of the appropriate institutional review board that approved the project should be mentioned. The International Journal of Pharmaceuticals, Nutraceuticals and Cosmetic Science reserves the right to request the related documents.
Manuscript Revision
The cover letter that accompanies your revised manuscript must include a point-by-point response to each comment by the reviewers and editor(s). Typically, this is addressed by listing each comment verbatim, and then directly following each comment with an explanation of how you addressed it, with reference to specific line or figure numbers in the revised manuscript. Using a different colour or font for the review comment vs. your response or inserting the review comments and your responses into a two-column table is strongly encouraged. Should you disagree with the constructive criticism raised, it is imperative that you provide a clear rationale for why you disagree in the point-by-point response.
Revised manuscripts and the point-by-point cover letters are submitted to ijpnacs@uitm.edu.my

